Delivering the Critical Factors for Clinical Confidence
The success of many clinical trials can hinge on the quality and timeliness of lab results. That’s why seven of the top ten largest CROs and prestigious medical research centers alike turn to NMS Labs for advanced testing services. From exclusionary pre-trial testing through post-marketing surveillance and new application for approved drugs, NMS Labs can help you and your clients make faster, more confident decisions at every phase of your study.
When responsiveness, customization, and scientific expertise are critical factors, you will work directly with one of NMS Labs’ experienced scientists to ensure the study parameters meet your unique needs.
Scientific Expertise
NMS Labs possesses a unique expertise that makes us an invaluable partner for your testing needs all the way up to your most complex, high-profile drug trials. Our 200+ scientific staff is comprised of leading board-certified toxicologists and clinical chemists with experience covering advanced bioanalytical, diagnostic, toxicology, and biomarker testing.
Data Quality and Integrity
- Accredited and meets rigorous standards of industry organizations such as ABFT, ANAB, ISO 17025, CLIA, CAP ISO 15189, CAP-LAP and NYS DOH
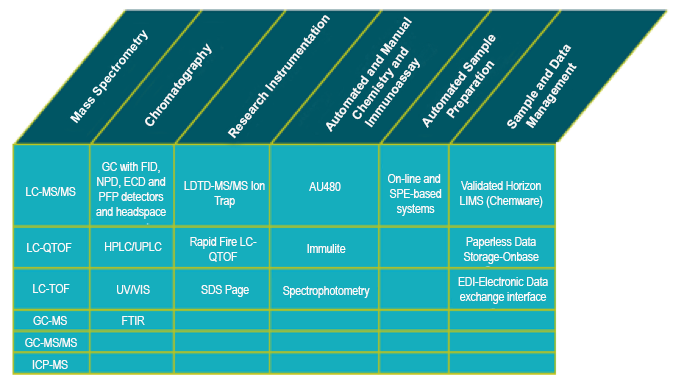
- Validated Horizon LIMS (Chemware) for sample and data management
Responsive and Customized Service
- Proposal requests are responded to within 24 hours
- Develop methods and customized reports to meet the specific demands of each client and trial
- Personalized Project Management
Customized Service and Reporting
- Develop customized methods and reports to meet the specific demands of each client and trial
- Web portal for real-time data access
- Experienced in providing Electronic Data Interface to national reference laboratories and clinical research organizations
Innovative Technology
We utilize state-of-the-art technologies and methodologies such as LC-MS/MS, ICP/MS, cell-based assays, and the highest-quality instrumentation available.
Flexible
Our capabilities cover unique sample types including tissues and all biological fluids.
Partner with NMS Labs Today
With NMS Labs as your partner, you can keep your trial moving forward cost effectively and more efficiently.
Call 1-866-522-2206 or email us at researchstudies@nmslabs.com any time to connect with one of our highly respected board-certified toxicologists and clinical chemists about your current or upcoming trial.